- Hangzhou firm advances retinal and cortical brain-computer interfaces
- Funding comes as China steps up support for neural technologies
Brain-computer interface (BCI) startup Nanochap (暖芯迦) has raised 300 million yuan ($44 million) in a strategic funding round, as the company steps up efforts to commercialize neural technologies aimed at restoring vision to blind patients.
The Hangzhou-based company said the round was co-led by venture capital firm Legend Capital and an undisclosed industry-focused investor, with existing backer Harvest Capital increasing its stake. GF Xinde Investment Management and Caitong Capital also participated.
The proceeds will be used to advance clinical trials for visual brain-computer interface products and upgrade core technologies.
The financing comes as brain-computer interfaces gain policy backing in China under the country’s 15th Five-Year Plan (2026-2030), with regulators and local governments accelerating work on technical standards, clinical approvals and industrial supply chains.
Meanwhile, visual reconstruction has emerged as one of the most closely watched areas globally in brain-computer interface development.
Neuralink has said its Blindsight device could help blind patients regain low-resolution vision as early as 2026, while Science Corporation released clinical data for its PRIMA visual prosthesis program last year.
The unmet demand remains immense. According to the World Health Organization, around 1.2 billion people globally live with some form of visual impairment, including 43 million blind individuals and 238 million with low vision.
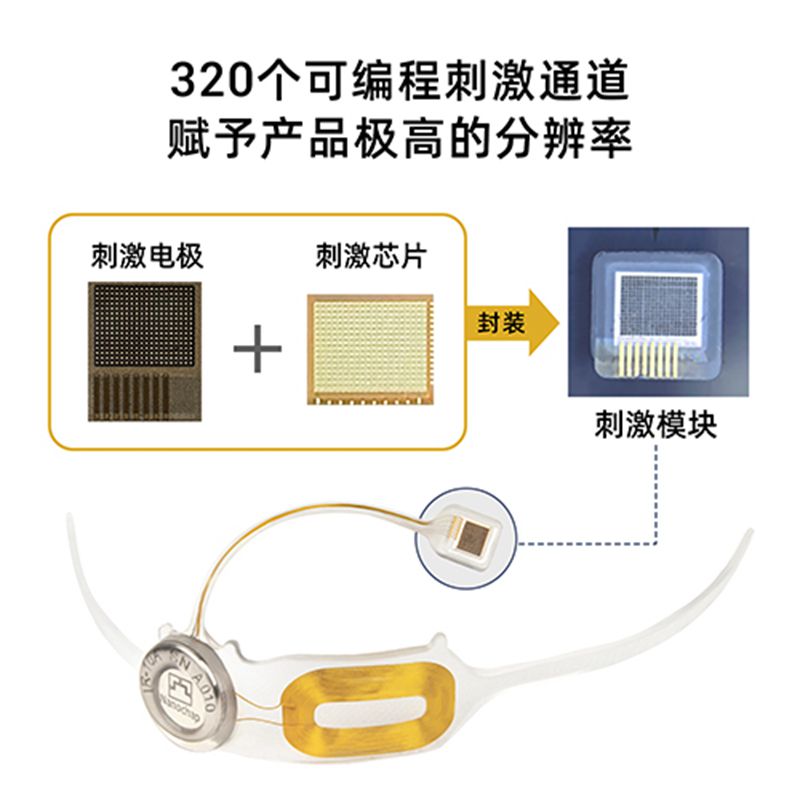
Nanochap is developing both retinal-pathway and visual-cortex brain-computer interfaces, branded as E-BCI and V-BCI.
The company said its systems now support as many as 1,280 recording and stimulation channels, allowing simultaneous monitoring and intervention across large numbers of neural regions.
Its retinal device uses minimally invasive eye surgery to implant electrode arrays into the macular region of the retina, stimulating retinal ganglion or bipolar cells to restore functional vision.
The approach has accumulated nearly a decade of clinical safety data globally and carries lower surgical and ethical risks than intracranial implants, potentially allowing commercial clinical deployment within two to three years.
Additionally, the visual-cortex approach implants electrodes directly into the brain’s visual cortex, bypassing damaged optic nerves entirely and expanding use cases to patients with missing eyes or optic nerve atrophy. That also raises substantially higher safety requirements because of the need for cranial surgery.
Prospective clinical trials
Nanochap’s retinal implant received type inspection certification from China’s Nationl Medical Products Administration in 2024, with the company saying tests confirmed biocompatibility and implant durability exceeding 10 years.
It has also completed animal trials involving nearly 100 large animals and primates.
Human clinical trials for the retinal implant are already underway, while the company plans to begin prospective clinical trials this year for its cortical interface product.
Nanochap said all core technologies—including electrodes, chips and packaging processes—were developed in-house, though it did not provide third-party verification for claims that its performance metrics rank among the world’s leading systems.


起飞-2.jpg)
